One of the key tools for combatting the opioid overdose epidemic, which claimed more than 100,000 lives in the last year, is the use of medications for opioid use disorder (MOUD). MOUD is a treatment approach that uses medications to relieve withdrawal systems or, in the event of a relapse, block the effects of opioids. MOUD is a vital tool to help individuals dealing with opioid use disorder (OUD) manage their recovery, and is considered the gold standard for treatment.
The three FDA-approved medications used in MOUD are methadone, buprenorphine (either as a solo formulation or with naloxone), and naltrexone.
Buprenorphine functions as a partial opioid agonist that causes the reduction of opioid withdrawal symptoms and cravings in OUD patients. Buprenorphine can also block the effects of other opioids, which supports recovery and reduces overdose risk. Buprenorphine’s approval to be prescribed or dispensed in an office setting differs from methadone, which can only be provided within more heavily regulated opioid treatment programs (OTPs).
Research shows that agonist medications can reduce mortality by up to 50% for people with OUD. Efforts to remove legal barriers to expanded buprenorphine access have focused primarily on the federal level, including the successful repeal of the X-Waiver in December 2022. However, state-level regulatory barriers to buprenorphine treatment have received comparatively little attention.
As a Schedule III controlled substance, however, buprenorphine is still subject to heightened regulation that can affect access to treatment. The regulations vary state to state and typically impact access in one of two ways: laws that affirmatively expand access to buprenorphine, and those that create high-threshold regimes within which providers and patients must clear legally imposed barriers to access buprenorphine. Ohio, for example, requires that office-based opioid prescribers incorporate counseling in their treatment plans, unless an exception is documented in the patient’s record. This can impede access for patients who may have difficulty making office visits or who may be reticent toward counseling.
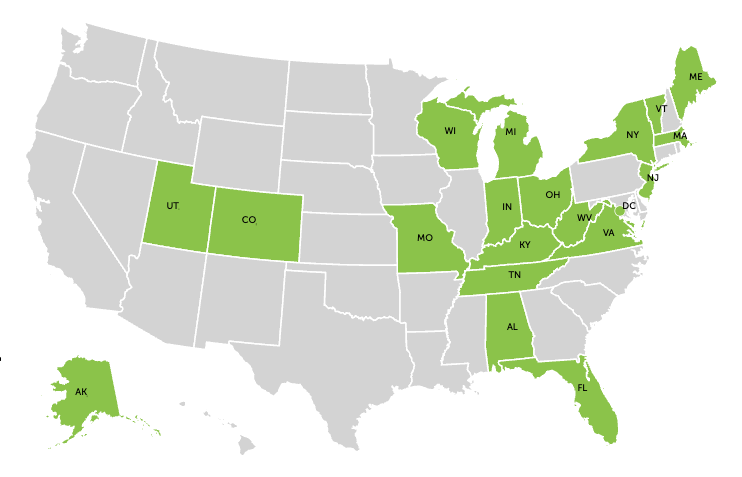
New data published by CPHLR with Vital Strategies Overdose Prevention Program now capture key features of state-level statutes and regulations regarding the prescribing of buprenorphine for OUD outside of federally certified opioid treatment programs. The dataset includes laws across all 50 states and the District of Columbia in effect as of March 1, 2023.
The data show
- Buprenorphine is generally regulated as a controlled substance in every state, but at least 19 states and the District of Columbia have laws that specifically target the prescribing of buprenorphine for OUD.
- Eight states and the District of Columbia established visit frequency requirements for some or all patients prescribed buprenorphine.
- Despite research showing that mandated counseling may inhibit treatment access, nearly one-third of states (14 states and the District of Columbia) had laws that mandated counseling had laws that mandated counseling for some or all buprenorphine patients. Of these, four states established a minimum frequency of counseling services, and nine states and the District of Columbia regulated the format or content of the counseling.
- Laws in 15 states incorporated outdated provisions of federal law related to the X-waiver.
- Only five states had laws that explicitly prohibited or discouraged patient discharge for specified conduct such as declining counseling services or positive drug screen results. Only one state —West Virginia — explicitly prohibited punitive reductions in prescribed medication dosages.
Research has shown that low-barrier buprenorphine treatment not only expands access to more patients, but it does so equitably. CPHLR researchers have identified four opportunities for state policymakers to improve access to care in a policy brief released with the data. More details on these recommendations and a research agenda for legal epidemiology may be found in the policy brief focused on this topic.
Access the data and more information on PDAPS.org.